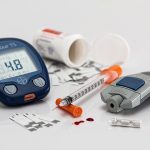
Microscopic view of the gelatin structure used to 3-D print a bioprosthetic mouse ovary implant. Credit: Northwestern University
Ten to 15 percent of couples in the United States are infertile and many are desperate to have children. That’s why a successful study a few months ago from researchers at Northwestern University‘s Feinberg School of Medicine and McCormick School of Engineering on bioprinting ovaries was so exciting. The team led by Teresa Woodruff, reproductive scientist and director of the Women’s Health Research Institute at Feinberg, and Ramille Shah, assistant professor of materials science and engineering at McCormick and of surgery at Feinberg, not only 3D printed ovaries, but implanted one of them in a mouse that then gave birth to healthy babies. The mouse’s natural ovary was removed and implanted with the 3D bioprinted ovary using a new gelatin like hydrogel (photo) the team invented to create the scaffold for the 3D printed organ. The mouse was able to ovulate with the artificial ovary, and once mated, she became pregnant and gave birth to healthy baby mice. The mouse mom was even able to lactate and nurse her babies. This great achievement in regenerative medicine advances us to a day not so far from now when we will be able to 3D print complex functional organs and transplant them into humans.
During the study Adam Jakus, a postdoctoral fellow was working as part of the 3D bio-printed ovary team, was preparing 3D printing ink made from ovarian cells. He spilled some of the solution in a fortuitous accident and before he could clean up, the ink solidified, forming a sheet of dry paper like material.
“During my initial attempts to create a 3D-printable formulation made from decellularized ovaries, I spilled a little of the material,” said Adam Jakus. “The spill rapidly dried into a robust sheet that could be picked up and easily handled. This gave me the idea to pursue this route, and I made similar ‘tissue papers’ from liver, kidney, uterus, muscle, and heart tissues and organs.”
The tissue paper which is pliable enough to bend and shape maintains proteins found in natural organs. Therefore immune cells in the body recognize and accept the paper once it’s implanted. Since each paper is made with proteins and biochemicals found in the specific organ it is derived from, each sheet is meant to be used in a particular place in the body.
“Each tissue paper type has different applications,” Jakus said. “For example, we are pursuing the use of the muscle tissue paper as a means of repairing or regenerating damaged or missing fine muscle tissue, such as in the face. Another example of use is for the ovary tissue paper to be used as means to maintain the life and function of ovarian tissue and follicles — the female egg unit — in vitro for the purposes of later transplantation.”
The paper detailing the research was published this week in the journal Advanced Functional Materials.
Video Transcript Below:
So we’ve created a material we call tissue papers that’s very thin like
0:20
phyllo dough made up of biological tissues and organs. We can switch out the
0:26
tissue that we use to make the tissue paper, so whether that be derived from
0:29
liver, or muscle or even ovary, we can switch it out very easily and make a
0:35
paper out of any tissue or organ.
0:41
A lot of people don’t want to buy kidneys, livers or hearts to eat,
0:44
so what the butchers do is just put them in bins in
0:47
the corner for very cheap. And that’s where we go and we collect our five, 10 hearts
0:51
or four or five livers and kidneys. And that’s what we’re use for the “de-cell” (decellularization)
0:54
process and make tissue papers.
1:08
So what this is showing is this heart piece here has been de-celled
1:11
one day, two day, three day, four day and five day. And you see it becomes clear.
1:16
This is a cow ovary. So it’s about the same size as a human
1:19
ovary. After you take out all the cells in the water, this is what ovaries are
1:23
like and it’s really what all our issues and organs would be like.
1:26
So we take our decellularized tissue or organ and we grind it into a fine powder.
1:29
And it’s that powder which we use to make the tissue papers.
1:32
Because it has components that are found in natural tissues and organs, the cells will
1:38
recognize it once it’s implanted.
1:40
What I’ve learned working here at
1:41
Northwestern a lot, especially in conjunction with Feinberg, working with
1:44
other surgeons, is that they care about how materials feel and handle. So as I
1:48
was picking this up, I was like, “I like how this feels,
1:50
it handles nice, it’s made of bioactive materials. There could be something here.”
1:53
Ultimately we’d want to create some type of patch or a very sophisticated
1:59
Band-Aid in order to regenerate damaged parts.
2:13
The follicles cells, or the ovarian cells, grew very well on it and they were
2:19
able to produce estrogen and so we think using this material in combination with
2:23
their ovarian cells could restore estrogen function.
2:27
It really expands what we can do when it comes to creating synthetic environments or synthetic
2:33
materials for tissue engineering applications.